The "Tripledemic of 2022" is Already Ending. Pfizer is Using it to Push its New RSV Vaccine for Pregnant Women
Is Pfizer Fanning the Hysterics Behind the Scenes?
The “tripledemic” of 2022, with flu, Covid, and RSV going simultaneously, is all over the news. Everyone, including myself and other substackers, is incentivized to play it up and catastrophize it.
Antivaxxers love talking about vaccine-destroyed immune systems.
Mask fanatics talk about… guess what… needing masks.
Mask haters explain how masks and lockdowns caused immune debt and/or unhealthy air for kids. Interestingly, fact-checkers promoting COVID vaccines agree with the “immune debt” theory while skirting around mask usage.
The CDC uses the “tripledemic” as an opportunity to promote Covid and flu vaccines.
The press loves vaccine-promotion grants from the CDC and dramatizing events to drive subscriptions.
Twitter neurotics love the opportunity to commiserate with similarly predisposed followers.
MedTwitter health experts dispense useless advice in hopes of increasing the follower count.
Pfizer is using the RSV wave of this year to drive up hopes for its poorly-tested and very ineffective future “RSV vaccine for pregnant women.” This is very important.
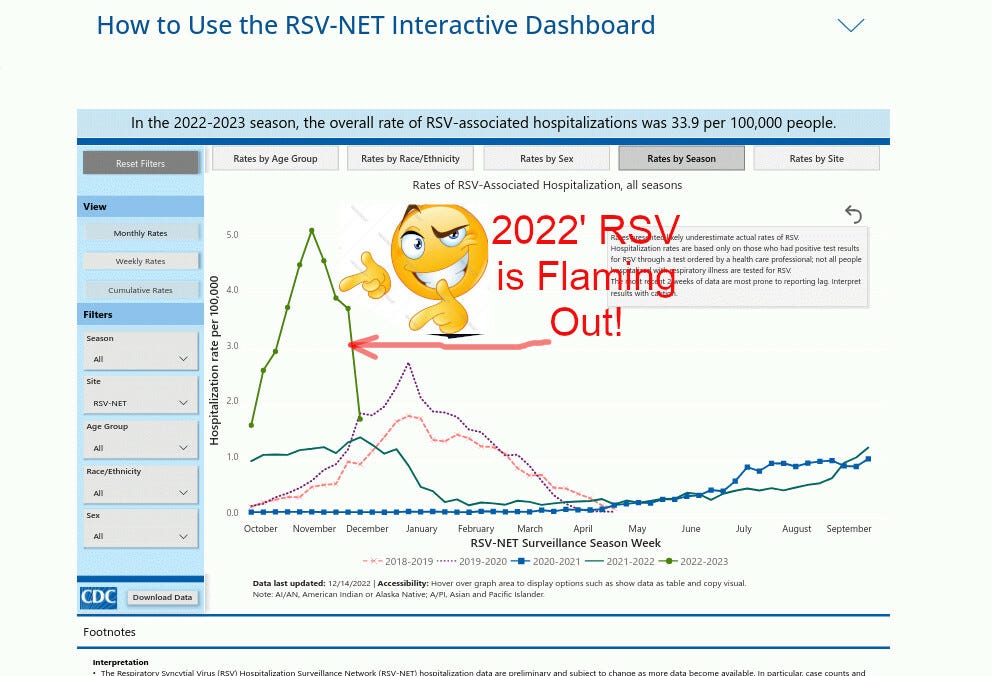
Let’s take a closer look at what is going on. Here’s the CDC’s RSV tracker:
It shows that RSV this season (green line) has been declining since a few weeks ago. I cannot predict the future, but chances are good that the total number of RSV cases will be in line with previous seasons, such as 2019, with lower peaks but longer duration.
CDC’s Influenza-like illness tracker also shows a decline in flu-specific tests:
It is possible that influenza and RSV waves this year were more pathogenic; we will not know this for some time until final statistics are collected. I believe that such an outcome is quite possible. Consider this influenza hospitalization (not incidence) chart from Canada:
For a very interesting take on the pathogenicity of flu and RSV in vaccinated people, read Geert Vanden Bossche’s Q&A No 25.
However, in terms of mere incidence and number of cases, this winter season is in line with pre-Covid years. A little shorter and a little steeper wave is all that is different.
The high influx of hospitalized patients happened partially because the wave of influenza and RSV was steeper (and shorter in duration) than usual. Think of this phenomenon as the opposite of “flatten the curve.”
RSV Fears Help Promote Pfizer’s Unproven RSV Vaccine
This Nature article is a typical specimen of Pfizer-RSV-vaccine-promoting fear porn omitting key information:
The article reads like an infomercial. So do most other press articles.
News media is sounding alarms at the “RSV emergency,” expressing certainty over the “high effectiveness” of Pfizer’s RSV vaccine, never questioning Pfizer press releases, and begging the FDA to expedite approval of this vaccine. Nature says:
In the past few months, the respiratory syncytial virus (RSV) has been sending children under the age of 5 to hospital at alarming rates in the United States and Europe. As paediatric units fill beyond capacity, and physicians contend with a ‘tripledemic’ of RSV, influenza and COVID-19, some have been calling for a state of emergency to be declared.
Pfizer reported in a press release that this strategy had an efficacy of 81.8% against severe cases of RSV in the infants for the 90 days after birth.
last week, the pharmaceutical company Pfizer announced that the US Food and Drug Administration (FDA) had agreed to review its RSV vaccine, to be administered to adults over the age of 60, as a priority. If approved — which might happen as soon as May — it could be the first sanctioned jab for RSV.
Nobody is asking, ”what is your emergency,” and nobody is suggesting to slow down a bit. Previous RSV vaccines, attempted decades ago, were also administered with few side effects. However, a year later, when vaccinated children faced RSV infections, 80% of the cohort of twenty vaccinated children ended up hospitalized, and two vaccinated kids died. This tragedy alone would warrant some caution, a longer review, and a gradual rollout.
The newborn infants in the Pfizer trial were followed for only 180 days.
The Pfizer RSV trial did not meet the predetermined endpoint (sad face below) and saw rapidly declining efficacy after birth:
Note: given the efficacy of 57% for the “first three months” and 51% for the “first six months,” I calculated the efficacy for “3-6 months” to be only 45%!
None of that matters to Pfizer, the FDA, and the Pfizer-paid press.
Pfizer's plans to vaccinate pregnant women using unspecified adjuvants should also subject the proposed injection to greater scrutiny.
However, Pfizer is asking for rapid approval, and the FDA seems agreeable to expediting Pfizer’s request. Why? Is the “emergency” itself mostly Pfizer-friendly-media hysterics, not supported by facts?
The clinical trial has no results posted — but the FDA is asked to approve the vaccine soon anyway. Sounds familiar?
Overplaying the RSV and influenza threat is in almost everyone’s interest — but the real beneficiary is Pfizer, which will use the fears to get rapid approval for an unproven adjuvanted subunit vaccine for pregnant women.
Is the RSV “emergency,” which benefits Pfizer’s RSV vaccine, caused by the Pfizer Covid vaccine? Is the “tripledemic” as terrible as the media claims it to be? Did you have any unusually severe colds this winter?







I would rather die here at home reading Igor's Newsletter than seeing the walls of a medical establishment.
And I am not alone with this sentiment...
There’s no replacement for the lack of trust. People seriously consider whether to go to the doctors that have failed them, leaving the government with desperate options like promoting assisted suicide.