FDA Advisers are ANGRY at Moderna for HIDING Data
Are they pretending to be independent to save themselves?
The times are changing!
The FDA’s “Independent advisers” are reportedly disappointed and angry at Moderna.
We, the antivaxxers, have been disappointed and angry at Moderna for about two or three years based on mountains of data we unearthed, some of which the FDA tried to hide for 75 years. So, may we ask, what disappointed the FDA advisers so late in the game? This is what it was:
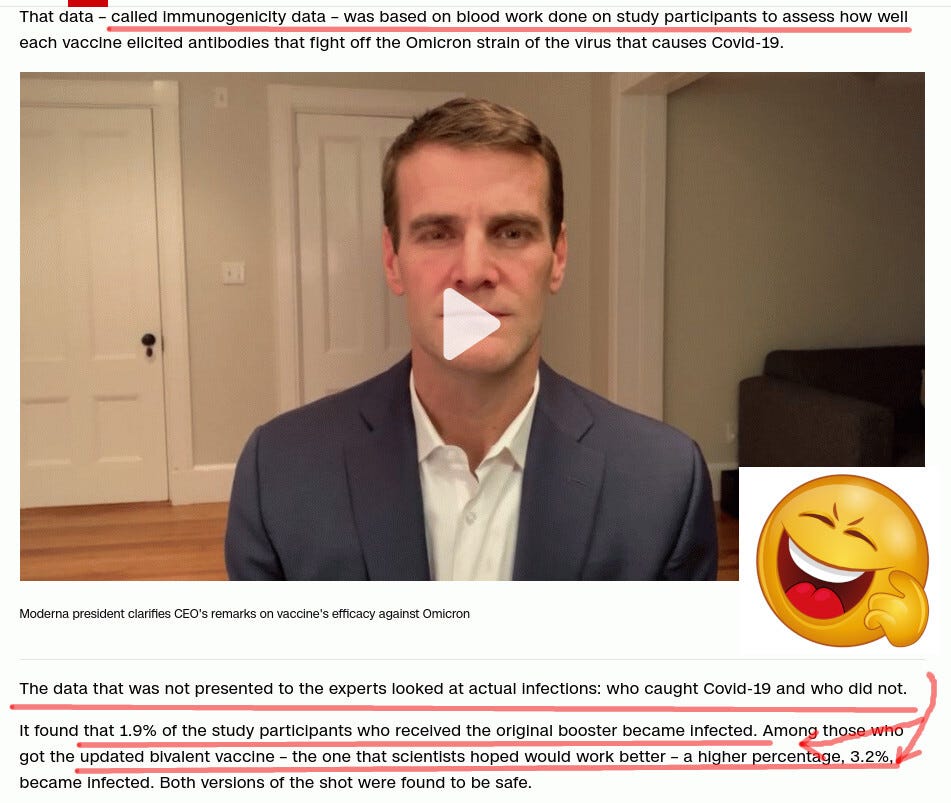
It turns out that the FDA advisers approved Moderna booster shots based on “antibody counting,” a quack medicine approach called immunobridging.
The reason immunobridging is medical quackery is pointed at by the “laughing emoji” above. Despite having “more antibodies,” MORE people in the bivalent group caught Covid compared to people in the monovalent (old booster) group.
Despite being there in larger numbers, the antibodies facilitated the infection instead of preventing it. Thus, reliance on antibody counts is medical quackery, as the real-life experimental data proves them useless at best.
FDA charlatans approved the Moderna bivalent Covid booster for millions of people based on antibodies in TEN MICE (who all got sick with Covid when challenged with the virus).
And now, the FDA advisers are “angry” that Moderna did not present real-life data showing that bivalent-boosted people are 68% (3.2/1.9) more likely to get Covid.
Why did they not get angry several months before?
The data that was not presented was available in plain sight!
Despite these imperfections, the data was included in a preprint study that was posted online in June, again in September in an FDA document and then later that month in a top medical journal – and advisers to the FDA and the CDC said the data should have been shared with them, too.
The FDA advisers are not reading the literature and top medical journals. What do they do in their spare time?
Michael Felberbaum, an FDA spokesman, told CNN in an email that “the FDA received the preprint less than a day prior to the advisory committee meeting,” and “the information was therefore not provided in an adequate timeframe for it to be included in the agency’s meeting materials, and generally the FDA only discusses data at advisory committee meetings that the agency has had the opportunity to substantively review.”
Hm. The FDA only discusses data at advisory committee meetings that the agency has had the opportunity to substantively review??? That’s a lie. The FDA approved the “Covid vaccine for babies” just TWO DAYS after receiving the submission.
The FDA was good at rubber stamping stuff while they considered themselves invincible.
Now that the public is asking pointed questions, FDA advisers are suddenly “angry” at Moderna. Are they trying to save themselves from repercussions and future prosecution?
Should we offer them “pandemic amnesty”?





Those of us with vaccine-injured children have known about this kind of data hiding and word-smithing for over a decade. We tried to warn people. It was all in plain sight. The idiots who spewed hate and posted dumb memes years ago about “if my kid can’t bring peanut butter to the classroom, yours can’t bring diseases” are now vaccine-injured and eating crow. Wakefield, Krigsman, Mumper, Megson, Kartzinel and many others have been sounding the alarm for decades. Now the clot shot is a whole other beast. CDC really broke out the big guns with this monstrosity, and MSM remains silent. They’ll keep going as long as they can. They don’t care if they injure or kill. It’s all about the Benjamins.
We are disappointed and angry that they failed to do their jobs. Hold them all accountable, especially Ashish Jha.